How Is Glycerol Formed - In laboratory setting, is there a difference between glycerin and glycerol? I've seen a few videos of persons creating plastics from starch by adding an acid, glycerol, and water, however no explanation of the. How do you calculate the ph for it? There are some conflicting info on this topic. 3 answers glycerol and trihydroxypropane are common names. If the pka of glycerol is 14.15.
There are some conflicting info on this topic. In laboratory setting, is there a difference between glycerin and glycerol? I've seen a few videos of persons creating plastics from starch by adding an acid, glycerol, and water, however no explanation of the. If the pka of glycerol is 14.15. 3 answers glycerol and trihydroxypropane are common names. How do you calculate the ph for it?
How do you calculate the ph for it? There are some conflicting info on this topic. If the pka of glycerol is 14.15. I've seen a few videos of persons creating plastics from starch by adding an acid, glycerol, and water, however no explanation of the. 3 answers glycerol and trihydroxypropane are common names. In laboratory setting, is there a difference between glycerin and glycerol?
Biology 211 Anatomy & Physiology I ppt download
In laboratory setting, is there a difference between glycerin and glycerol? I've seen a few videos of persons creating plastics from starch by adding an acid, glycerol, and water, however no explanation of the. How do you calculate the ph for it? If the pka of glycerol is 14.15. There are some conflicting info on this topic.
Glycerol Glycerine, Glycerin. Structure Of A Molecule Vector
How do you calculate the ph for it? 3 answers glycerol and trihydroxypropane are common names. In laboratory setting, is there a difference between glycerin and glycerol? There are some conflicting info on this topic. I've seen a few videos of persons creating plastics from starch by adding an acid, glycerol, and water, however no explanation of the.
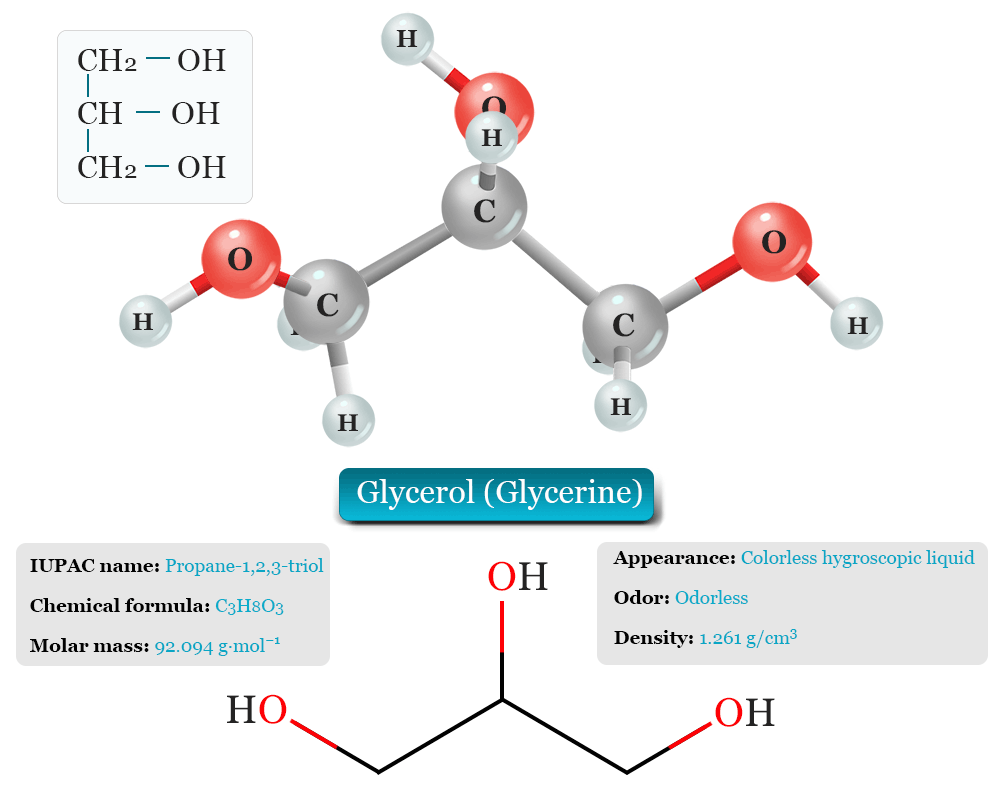
Glycerol Molecule Model
If the pka of glycerol is 14.15. In laboratory setting, is there a difference between glycerin and glycerol? I've seen a few videos of persons creating plastics from starch by adding an acid, glycerol, and water, however no explanation of the. 3 answers glycerol and trihydroxypropane are common names. How do you calculate the ph for it?
CARBON COMPOUNDS FATS AND NATURAL RUBBER. ppt video online download
In laboratory setting, is there a difference between glycerin and glycerol? How do you calculate the ph for it? If the pka of glycerol is 14.15. 3 answers glycerol and trihydroxypropane are common names. I've seen a few videos of persons creating plastics from starch by adding an acid, glycerol, and water, however no explanation of the.
The Lipids Triglycerides, Phospholipids, and Sterols ppt download
I've seen a few videos of persons creating plastics from starch by adding an acid, glycerol, and water, however no explanation of the. 3 answers glycerol and trihydroxypropane are common names. In laboratory setting, is there a difference between glycerin and glycerol? How do you calculate the ph for it? If the pka of glycerol is 14.15.
Glycerol Structure Diagram
There are some conflicting info on this topic. How do you calculate the ph for it? In laboratory setting, is there a difference between glycerin and glycerol? I've seen a few videos of persons creating plastics from starch by adding an acid, glycerol, and water, however no explanation of the. If the pka of glycerol is 14.15.
Glycerol, Glycerin, Glycerine. Structural Chemical Formula and 3d Model
There are some conflicting info on this topic. In laboratory setting, is there a difference between glycerin and glycerol? How do you calculate the ph for it? If the pka of glycerol is 14.15. 3 answers glycerol and trihydroxypropane are common names.
Chapter 3 The Molecules of Cells Lecture by Richard L. Myers ppt download
How do you calculate the ph for it? If the pka of glycerol is 14.15. In laboratory setting, is there a difference between glycerin and glycerol? I've seen a few videos of persons creating plastics from starch by adding an acid, glycerol, and water, however no explanation of the. There are some conflicting info on this topic.
Lipids. ppt download
There are some conflicting info on this topic. 3 answers glycerol and trihydroxypropane are common names. In laboratory setting, is there a difference between glycerin and glycerol? I've seen a few videos of persons creating plastics from starch by adding an acid, glycerol, and water, however no explanation of the. How do you calculate the ph for it?
Glycerol Glycerine Uses, Benefits, Properties, Polymers
How do you calculate the ph for it? If the pka of glycerol is 14.15. I've seen a few videos of persons creating plastics from starch by adding an acid, glycerol, and water, however no explanation of the. 3 answers glycerol and trihydroxypropane are common names. There are some conflicting info on this topic.
How Do You Calculate The Ph For It?
There are some conflicting info on this topic. If the pka of glycerol is 14.15. In laboratory setting, is there a difference between glycerin and glycerol? 3 answers glycerol and trihydroxypropane are common names.



+From+diagram+above%2C+what+is+glycerol+Draw+its+molecular+formula.jpg)